- Mon. – Fri.: 8:30 a.m. – 5:00 p.m.
Solution: Actions to Take in Case of Deviations
With the enaio® Best Practice Solution (BPL) for Deviation Management, which is part of a digital quality management system, deviation reports can be centrally recorded and processed through a workflow-driven process.
BPL’s Deviation Management provides all the necessary tools for efficient digital deviation management. The system focuses on workflow-driven tasks, comprehensive logging, and the creation of electronic files for the entire deviation process. All relevant information is digitally accessible at any time. In addition, the solution enables simple analyses to identify recurring issues and implement CAPA processes, thereby significantly increasing transparency and process reliability.
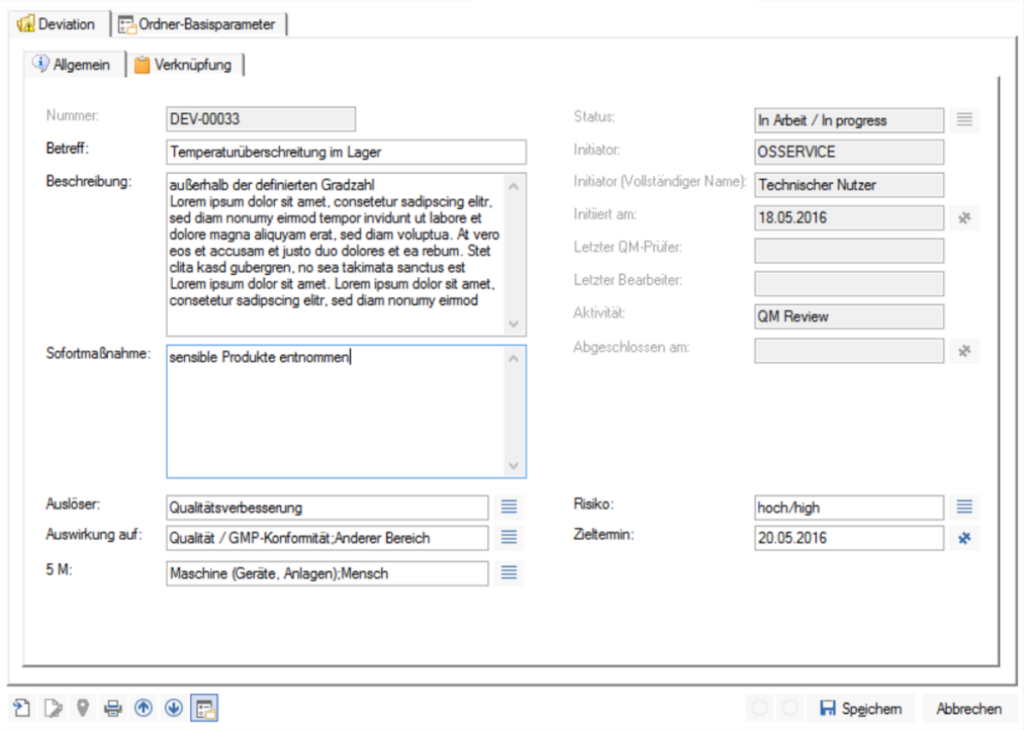
enaio® helps you address potential risk scenarios within your organization quickly and flexibly. Current status, department, trigger—the necessary information is immediately available.
Responding quickly to irregularities also means taking action, initiating the appropriate steps, and assigning responsibilities:enaio® workflows document the entire process—in a traceable and transparent manner.
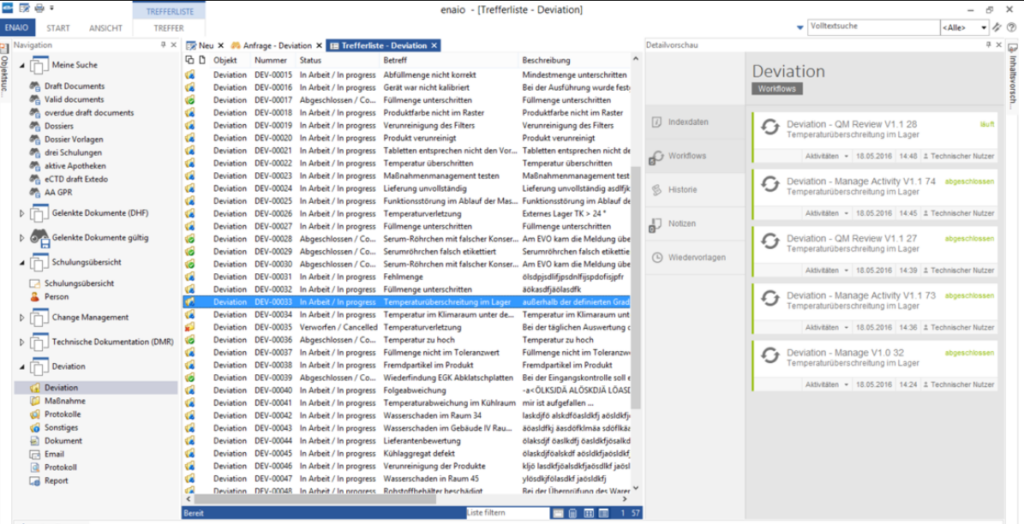
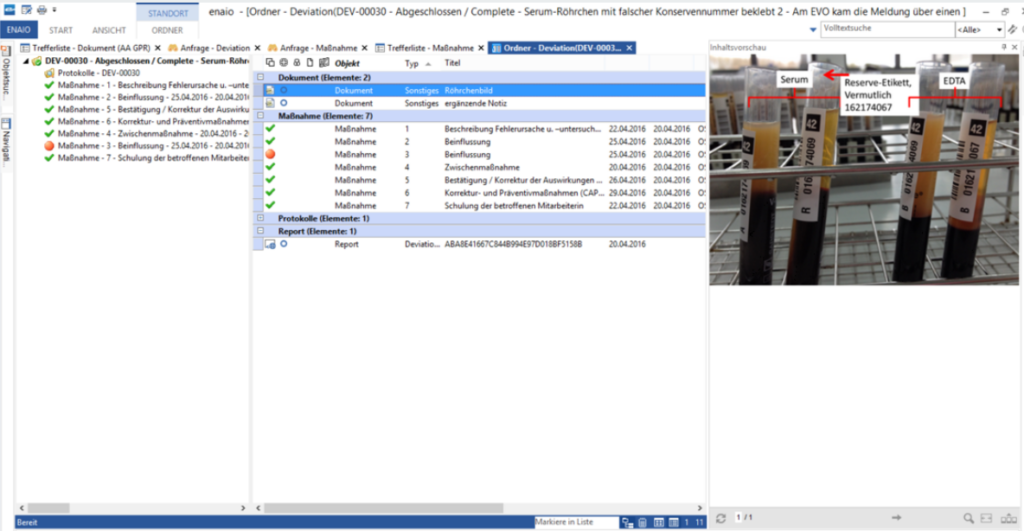
All relevant data and documents are stored in a central location. Ensure access anytime, anywhere to minimize administrative effort.
The increasingly prevalent approach of subjecting everything to a risk assessment is also incorporated into our Deviation/CAPA best-practice solution. However, the assessment and evaluation of a risk is always the responsibility of the individuals involved in the process. The enaio® best-practice solution serves as a tool to better handle, monitor, and document the necessary tasks.
When decisions regarding product quality are made based on data processed in deviation management, you must be able to rely on the integrity of that data. According to the recognized ALCOA concept, data must be accurate, legible, timely, original, and traceable. With enaio® best-practice solutions, you can be confident that you are maintaining regulatory compliance.